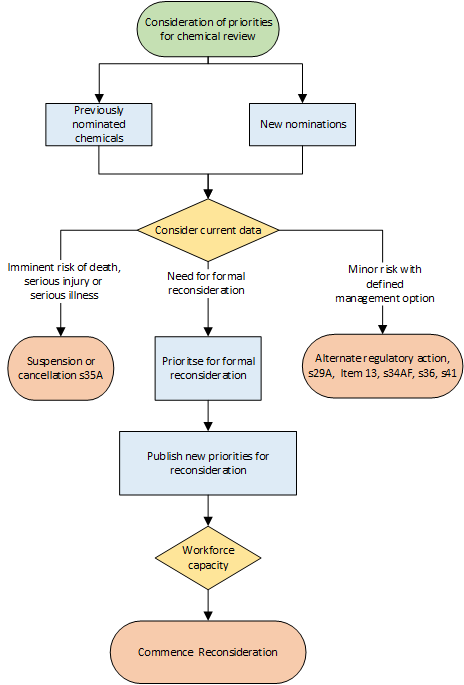
Chemicals nominated for reconsideration are assessed and prioritised based on the potential risk indicated by the available scientific information.
The priority reconsiderations listed by the Australian Pesticides and Veterinary Medicines Authority (APVMA) represents those products which have been identified as having the most significant potential risks and these reconsiderations are expected to be commenced within the next 5 years. This list is intended to provided transparency for stakeholders about the APVMA’s planned reconsiderations.
Other chemicals which have previously been nominated remain on the nomination list and will be considered in future prioritisation processes.
The APVMA continually monitors published scientific reports, international regulatory decisions and adverse experience reports and will take regulatory action on any chemical if there is an imminent risk of serious harm to people. If risk mitigation measures which would reduce the risk associated with the use of a product based on current knowledge are identified, the APVMA has regulatory mechanisms other than formal reconsideration available.
Regulatory mechanisms available to the APVMA to address concerns with registered products or approved active constituents and labels include:
- varying the label or conditions of approval or registration with the holder’s consent (section 29A)
- issuing an invitation to submit an Item 13 variation to address minor changes at no cost to the holder (Division 3, section 26 to 29)
- making mandatory variations to the label in certain circumstances (section 34AF)
- suspending or cancelling a registration or approval if a condition of registration or approval is breached (section 36)
- suspending or cancelling a registration or approval after giving notice to the holder, where it appears to the APVMA that products, active constituents or labels may not meet the statutory criteria (section 41)
- suspending or cancelling a registration or approval without notice, where the APVMA considers it necessary to do so to prevent imminent risk to persons of death, serious injury or serious illness (section 35A).
The APVMA prioritises agricultural and veterinary chemicals nominated for review according to the scientific evidence supporting the nomination. The key criteria for concern are scientific evidence of potential risks to human health (toxicology and occupational health and safety), environment, residues and trade, and target crop and animal safety and efficacy.
The prioritisation process is represented in the flow chart in Figure 1.
Figure 1: A visual representation of the APVMA’s process to decide the appropriate course of action for chemicals nominated for review
Criteria for prioritisation of chemicals for review
Chemicals nominated for review may be prioritised for formal reconsideration or subject to alternative regulatory measures. Formal reconsideration of chemicals is prioritised based on the evidence indicating potential risk and the level of concern. Potential risks are identified using screening level risk assessments of new and previously considered data, together with expert scientific judgment of the level of concern indicated by the data. Assessments conducted to prioritise reconsiderations are not intended to be complete risk assessments but are used to identify the need for reconsideration or other regulatory actions.
To determine the priority of reconsiderations, subject matter experts for each risk assessment area (human health, chemistry, residues, trade, environment, and efficacy and safety) assign a score to the chemical depending on the type of evidence, level of concern and potential risk. The combined result for each chemical is used to determine whether it is a priority for reconsideration. The list of chemicals prioritised for reconsideration is published on our website.
The chemicals which are not prioritised remain on the nomination list and alternative regulatory action may be taken to reduce or mitigate any identified risk if appropriate mitigation measures are clearly identified.
The APVMA will take immediate regulatory action on any chemical, if it is identified as an imminent risk for people of serious injury, serious illness, or death, regardless of whether it has been prioritised for reconsideration.